- Contact Person : Mr. Shao Sam
- Company Name : Qingdao Bestex Rubber & Plastic Products Co., Ltd.
- Tel : 86-532-84356677,13325010175
- Fax : 86-532-84356699
- Address : Shandong,Qingdao,No.8-5 Guangzhou Road, Pingdu, Qingdao, Shandong, China
- Country/Region : China
- Zip : 266700
Related Product Searches:FDA510K Certification Sterilization Pouch,High Quality,FDA510K Certification Sterilization Pouch, sterilized paper pouch,SSP-27510
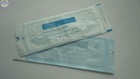
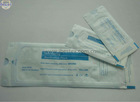
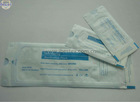
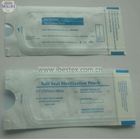
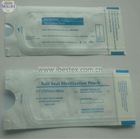
FDA510K certification sterilization pouch
Advantage
1) See interior items clearly through transparent film
2) Have good sterile barrier
3) Excellent peelability
4) Seal quickly
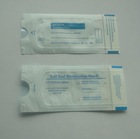
FDA510K certification sterilization pouch
Specifications
1. Adopt special double-sided tape to adhesive seal quickly and effectively without professional equipment.
2. Adopt three side explosion-proof technology to prevent burst effectively.
3. With sterilization change-color instructions to show sterilization conditions clearly.
4. See interior items clearly through transparent film
5. Adapt to sterilization methods: Ethylene oxide (ETO), pressure steam (STEAM)
* Initial blue turns yellow(EO GAS Sterilization)
* Initial pink turns brown(Steam Sterilization)
FDA510K certification sterilization pouch
Functions
All are used by dentists ,salons ,hospitals, spas, tattoo arttists and many other types of medical facilities to enable the sterilization and storage of surgical instruments, medical gloves and wond care suppliers prior to use.
General Size
FDA510K Certification Sterilization Pouch